Supplier Compliance for
Regulated Medical Products
VendorWeave automates MDR materials declarations, tracks ISO 13485 and FDA certifications, manages risk-based onboarding, and scores supplier performance — built for teams that answer to auditors.
See It In Action
Track ISO 13485, FDA, and MDR compliance in one dashboard
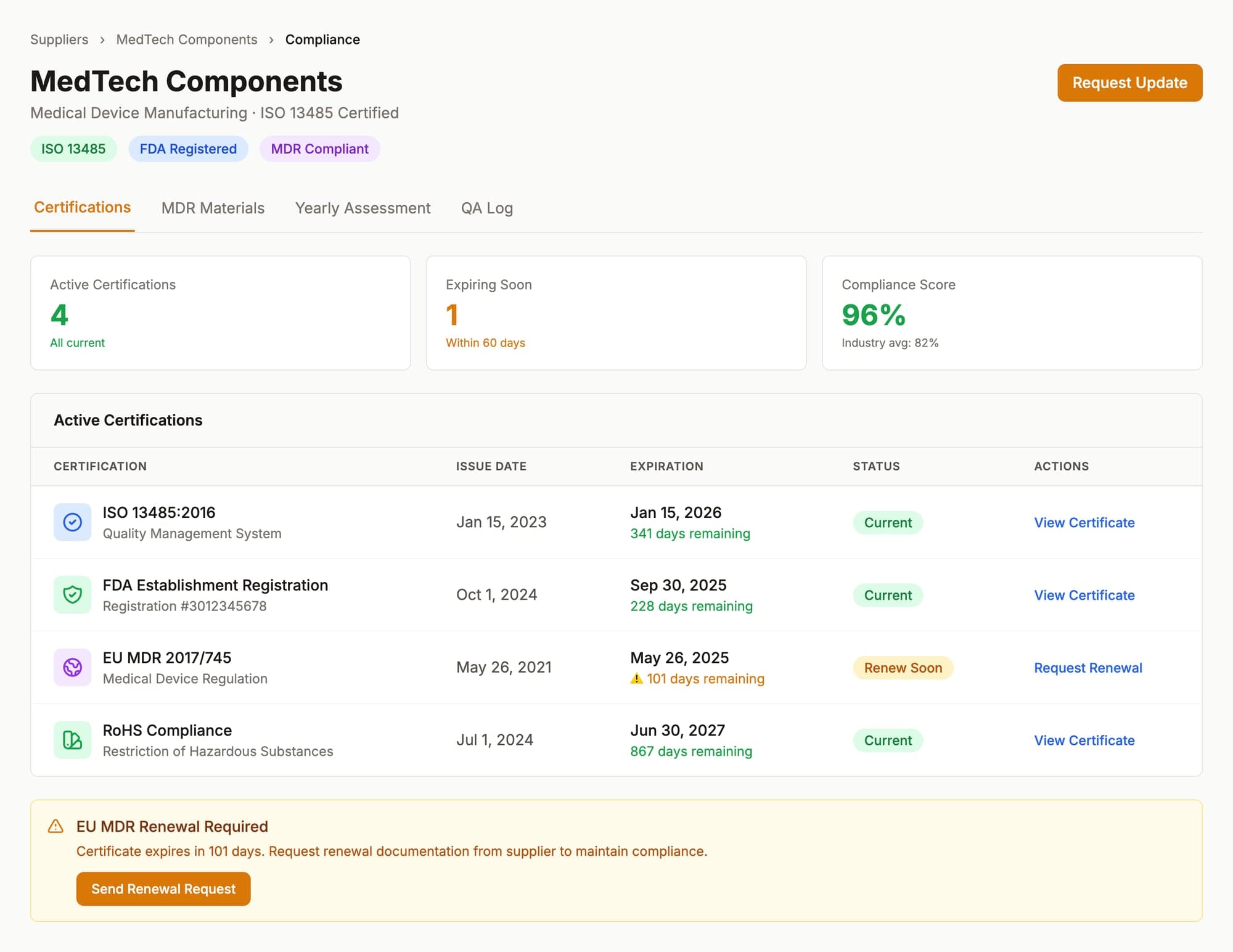
Real-time visibility into supplier certifications with automated expiration alerts
Built for Medtech Compliance Challenges
MDR materials declarations stuck in email threads for weeks
Automated MDR Annex I survey collection with AI parsing and deadline tracking
ISO 13485 and FDA registration certs expire without anyone noticing
10-day and 5-day expiration alerts with automated renewal requests to suppliers
Supplier onboarding takes 6+ weeks with manual document chasing
Risk-based onboarding workflows with auto-reminders — high-risk suppliers get the full package automatically
Quality events (CAPAs, complaints) scattered across spreadsheets and email
Centralized quality log with CAPA, SCAR, and complaint tracking tied to supplier scorecards
Regulatory Coverage
VendorWeave tracks the certifications and declarations your auditors ask for.
What Medtech Teams Use Most
Compliance Tracking
ISO 13485, FDA, MDR, RoHS, REACH — all in one dashboard with expiration alerts.
Supplier Scorecards
Five-metric scoring tied to quality logs. Know which suppliers are improving or declining.
Quality Management
CAPAs, SCARs, and complaints tracked per supplier with full audit trail.
Ready to automate medtech compliance?
Start free with 5 suppliers. All compliance features included. No credit card required.